Clinical research requires precision and streamlined processes, especially when it comes to managing complex tasks like billing, invoicing, and integrating multiple systems. Clinical research management software (CRMS) or clinical trials management system software (CTMS) plays a pivotal role in ensuring these tasks are handled efficiently, connecting study and financial teams seamlessly.
This software supports compliance and improves overall research management by enhancing communication and automating crucial workflows. Explore how this powerful software can transform your clinical research operations and improve efficiency – read on to discover more!
Benefits of clinical research management software
Implementing clinical research management software provides numerous advantages for organizations involved in clinical trials and research projects. This specialized platform streamlines research operations, improving productivity and coordination across teams, institutions, and study sites. By centralizing data and automating key processes, CTMS enhances the overall management of clinical studies, ensuring smoother and more efficient research workflows.
Below are key benefits of clinical trial management system:
Accurate and accessible data: Clinical research management software keeps essential data accurate, accessible, and up-to-date for ongoing trials. Its data visualization capabilities enable real-time tracking of study metrics, supporting more informed and effective decision-making.
Enhanced collaboration: CTMS is a collaboration platform, that allows team members to collaborate smoothly, even across remote or distributed teams, fostering more efficient teamwork.
Automation for efficiency: Workflow automation simplifies clinical trials management tasks, including progress tracking, patient record updates, and report generation. This improves productivity while reducing the need for manual effort.
Ensuring research integrity: Software ensures the integrity of clinical trials by enforcing transparency and regulatory compliance, promoting adherence to research ethics and proper procedural guidelines.
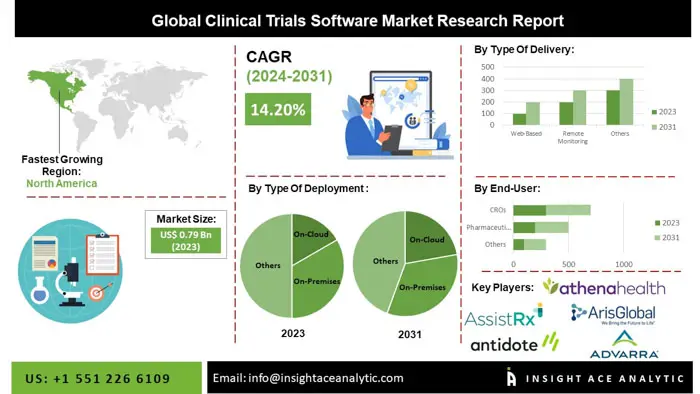
Global Clinical Trials Software Market to grow at 14.2% CAGR by 2031
The role of clinical trial management software in enhancing collaboration
Clinical Trial Management Software (CTMS) is pivotal in fostering collaboration among research teams by offering a unified, efficient platform for communication and data management. Here’s how CTMS improves team work:
Centralized data access: Clinical trial software offers real-time data access to all team members, promoting collaboration and minimizing information gaps between departments.
Streamlined communication: Built-in messaging and notification systems enable quick updates on trial progress and protocol changes, facilitating efficient communication.
Task management and tracking: CTMS assigns tasks, sets deadlines, and tracks progress, ensuring transparency and accountability.
Document version control: CTMS ensures team members stay informed about the latest protocols and documents by maintaining version control, minimizing errors and avoiding miscommunication.
Integration with collaboration tools: Advanced CTMS links with widely used platforms, facilitating streamlined workflows and effective information exchange among cross-functional teams.
Integration capabilities: CTMS connects systems for seamless document management and remote monitoring, ensuring smooth data sharing across platforms.
Collaborative features: Real-time updates on patient engagement and trial progress enable teams to collaborate effectively and make timely decisions.
By enhancing collaboration, CTMS drives efficiency and accelerates decision-making, ultimately improving the success of clinical trials.
Key components of clinical research management software
Advanced clinical research management software combines key tools to simplify and optimize the complex workflows involved in clinical trials. Here are the key components:
Study startup tools: These tools expedite trial initiation by automating regulatory document tracking and compliance tracking with approvals, such as IRB and FDA submissions.
Document management systems: Functions such as an electronic Trial Master File (eTMF) help organize trial documents, enforce version control, and provide secure, role-based access to confidential information.
Electronic data capture (EDC): Integrated EDC systems allow real-time data entry into electronic case report forms (eCRFs), enhancing data accuracy and shortening validation timelines.
Financial management tools: Budgeting and payment automation features track expenses, forecast costs, and ensure timely payments to research sites, enhancing financial accuracy and efficiency.
Reporting and dashboards: Customizable dashboards offer real-time insights into key performance indicators (KPIs) like patient enrollment and milestone progress, empowering informed decision-making.

Key components of clinical research management software
These components collectively streamline trial management, ensuring compliance, efficiency, and high data quality for successful clinical research.
Difference between CRMS, Electronic data capture and Excel
Clinical research management software vs Electronic data capture
Clinical research relies on robust systems to manage various aspects of research. Two essential tools in this process are the Clinical research management software and the Electronic data capture (EDC). While both serve critical roles, they complement each other, with CTMS focusing on project management and EDC centered on patient data collection.

Clinical research management software often integrates these systems to streamline operations. Including these below areas:
Tracking anonymized subject records, allowing study teams to monitor high-level screening, screen failures, and enrollment data. The software helps track participant enrollment, enabling the study management team to monitor progress, manage site payments, and create visit reports efficiently.
Subject visit progress, tracking visit dates and statuses.
Data collection, ensuring data is reviewed, verified, and approved efficiently.
Recording the end-of-study disposition, such as whether subjects completed or discontinued the study, along with the corresponding dates.
Clinical research management software vs Excel
Worksheets and spreadsheets are commonly used for clinical research tracking due to their simplicity and ease of use. However, as clinical teams grow and the complexity of trials increases, worksheets quickly reveal their limitations.

In contrast, CTMS offers several benefits that help organizations manage studies more efficiently and compliantly, including:
Collaboration with user roles: CTMS offers a secure environment that enables efficient teamwork among team members. User roles are controlled, ensuring that access to sensitive data is restricted based on the user's permissions and role within the study.
Integrated, consistent data: With CTMS, data consistency across different studies and views is ensured. The system centralizes information, allowing data to be applied uniformly across all trials, reducing errors and discrepancies.
Secure and reliable: It guarantees secure access, restricting data visibility to authorized users only. The system leverages cloud technology, with built-in backups and redundancy, guaranteeing data availability whenever it is needed.
Regulatory compliance: One of the key advantages of software is its automatic enforcement of regulatory requirements, such as 21 CFR Part 11 compliance. This includes features like audit trails, electronic signatures, and data archiving, all seamlessly integrated into daily operations.
Most common CRMS integrations throughout the enterprise
A clinical research management system serves a crucial role in the clinical research process. To enhance this functionality, many research sites choose to integrate their CTMS with other enterprise systems. This integration enhances workforce productivity, ensures data precision, and strengthens patient safety.

Below are some most common CTMS integrations:
Protocol and subject information: By implementing the IHE HL7 Retrieve Process for Execution (RPE) protocol, software can transfer critical information to an EMR system. It also enables the transfer of subject data, allowing enrolled research participants to be identified within the EMR system.
Demographics: The patient demographics interface automatically transfers essential details, such as contact information and date of birth, from the EMR system. This saves time and minimizes the risk of human error during manual data entry.
Protocol billing grid: This vital interface allows research teams to define billing assignments for procedures at the protocol level. The assigned data is synced with the Electronic Medical Record (EMR) system, facilitating accurate charge routing. This integration ensures compliance with billing standards while enhancing communication between clinical and financial teams.
General ledger: By linking the CTMS with a site's general ledger system, this interface allows for seamless management of CTMS-generated invoices. Payments for research activities can be reconciled in the software, providing research and financial teams with accurate, up-to-date information on study-related invoices.
Other research systems: Comprehensive software typically connects with additional platforms, including eRegulatory, eIRB, and participant payment systems. While native integrations may not always exist, Application Programming Interfaces (APIs) can facilitate connectivity, ensuring streamlined operations across various platforms.
How to choose the right clinical research management software?
While many modern clinical trial management systems offer similar features and capabilities, selecting the right one requires thoughtful evaluation. It’s essential to choose a system that can adapt to the ever-changing landscape of clinical trials and research.

These criteria cannot be neglected below:
Customizability: A strong clinical trials software should provide extensive customization options, especially for report generation and process automation. It must also align with the organization’s standard operating procedures and internal policies. This flexibility is essential to tailor the software to specific clinical trial needs.
Usability: While managing clinical trials can be intricate, CTMS itself should not be overly complex. A user-friendly interface is crucial, especially for teams adopting it for the first time, ensuring smooth onboarding and ease of use.
Accessibility: With research teams often spread across multiple locations, the ability to access the software remotely is essential. Web-based solutions provide the flexibility needed for efficient collaboration, ensuring that team members stay connected and informed, regardless of their location.
Interoperability with other systems: Clinical research management software should integrate seamlessly with existing tools and systems in use by your organization. This ensures that data can flow smoothly between platforms, enabling better process automation and reducing the risk of errors in data migration.
Value for money: Price points can vary significantly depending on the software's deployment options and available service packages. It's important to choose a platform that fits within the budget while still addressing the specific needs of the clinical trials and the organization.
Challenges in developing clinical research software
Legacy CTMS has been in use for nearly two decades, and despite its outdated nature, many organizations continue to rely on this software. It’s unexpected that many organizations lack immediate plans to update their clinical trial management processes, including their software systems. However, this presents a significant challenge. The evolving demands of modern clinical trials require advanced tools that can keep pace with regulatory requirements, data complexity, and operational efficiency.
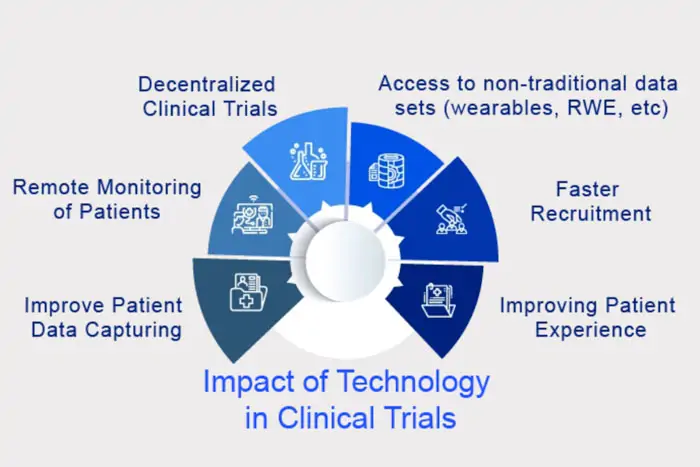
With most legacy clinical site startup tools still relying on on-premise deployments, they fall short in supporting decentralized clinical trial procedures. Transitioning to cloud-based clinical research management software appears to be the logical step forward, yet this shift entails a significant transformation of established workflows. This requires retraining staff, redefining roles, and creating a new procedural framework.
Another critical challenge is the interoperability between various systems. Studies examining clinical operations highlight that the primary obstacle is the integration of clinical applications - an area where legacy CTMS underperforms. Thus, its efficiency should aim to address these challenges effectively.
In today’s fast-evolving healthcare environment, finding effective solutions to enhance clinical research is more important than ever. With experience in working with leading healthtech companies and universities, TMA Solutions is dedicated to delivering practical tools for clinical research.
Partnering with TMA Solutions for developing clinical research tools means tapping into a wealth of experience, innovation, and cutting-edge technology tailored specifically for the healthcare sector. With 17+ years of experience and a team of over 600 engineers, TMA Solutions delivers dependable and advanced solutions. Their expertise enables them to address the complex demands of clinical research effectively.
TMA Solutions' dedication to quality and innovation keeps clinical research tools at the forefront of technological advancements. This leads to more efficient data management, streamlined research workflows, and better decision-making capabilities. TMA’s systems integrate smoothly with Health Information Systems (HIS) and Electronic Medical Records (EMR), enabling efficient data sharing. This ensures clinical studies and research have real-time access to accurate patient information.
Additionally, TMA’s expertise in healthcare scheduling systems and patient service platforms fosters greater coordination between research personnel and participants. TMA Solutions streamlines clinical research processes, from managing trial appointments to enabling real-time health monitoring. This enhances participant experiences while delivering better research outcomes.
Choosing TMA Solutions means benefiting from their deep expertise in healthcare technology and innovative solutions. Their clinical research tools improve research quality, accuracy, and efficiency, driving better health outcomes and streamlined healthcare delivery.
In summary, clinical research management software is an essential tool for simplifying operations, enhancing accuracy, and fostering better collaboration between research and financial teams. Its ability to integrate various systems and automate key processes ensures smoother workflows and improved compliance across clinical studies. For more insights on how this software can elevate your research efficiency, stay tuned for updates and information about TMA Solutions' cutting-edge innovations in the field.



